Summary
Midazolam is frequently used to produce sedation in critically ill pediatric patients. We studied the pharmacokinetics of midazolam in 22 patients (age 8 days to 16 years). The intravenous infusion rate to produce sedation ranged from 49–385 mcg/kg/hr.
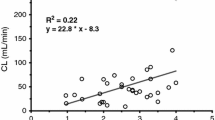
The blood samples were obtained at steady-state and midazolam was measured by gas chromatography with electron capture. The steady — state plasma concentrations of midazolam ranged from 49–385 ng/mL. The total clearance, apparent volume of distribution, and elimination half — life ranged from 0.1–3.1 L/kg/hr, 0.2–3.5 L/kg, and 0.3–10.9 hours, respectively. The marked interpatient variability in pharmacokinetics explains in part, the substantial variation in dosage requirements of midazolam to produce sedation in critically ill pediatric patients.
Similar content being viewed by others
References
Blumer JL. Clinical pharmacology of midazolam in infants and children. Clin Pharmacokinet 35: 37–47, 1998.
Nahata MC. Pharmacokinetics of doxacurium during normothermic and hypothermic cardiopulmonary bypass surgery. Can J Anaesth 45: 515–20, 1998.
Allonen H., Ziegler G., Klotz, U.. Midazolam kinetics. Clin Pharmacol Ther 30, 653–61, 1981.
Vasiliades J., Sahawneh T., Midazolam determination by gas chromatography, liquid chromatography and gas chromatography — mass spectrometry. J Chromatogr 228: 195–203, 1982.
Lee TC, Charles BG, Harte GJ, Gray PH, Steer PA, Glenady VJ. Population pharmacokinetic modeling in very premature infants receiving midazolam during mechanical ventilation: midazolam neonatal pharmacokinetics. Anesthesiology 90: 451–7, 1999.
Payne K., Nattheyse FJ, Liebenberg D., Dawes T., The pharmacokinetics of midazolam in paediatric patients. Eur J Clin Pharmacol 37: 267–72, 1989.
Jacqz-Aigrain E., Wood C, and Robieux I. Pharmacokinetics of midazolam in critially ill neonates. Eur J Clin Pharmacol 39: 191–2, 1990.
Jacqz-Aigrain E., Daoud P., Butrin P. Maherzi S., Beaufils F., Pharmacokinetics of midazolam during continuous infusion in critically ill neonates. Eur J Clin Pharmacol 42: 329–32, 1992.
Hartwig, S., Roth B., Theisohn M., Clinical experience with continuous intravenous sedation using midazolam and fentanyl in the paediatric intensive care unit. Eur J Pediatr 150: 784–8, 1991.
Tolia V., Brennan S., Aravind, MK, Kauffman, RE. Pharmacokinetic and pharmacodynamic study of midazolam in children during esophagogastro — duodenoscopy. J. Pediatr 119: 467–71, 1991.
Burtin T., Jacqz-Aigrain E., Girard P., Lenclen R., Magny JF., Betremieux P., Tehiry C., Desplanques L., Mussat P., Population pharmacokinetics of midozolam in neunates. Clin Pharmacol Ther 56: 615–25, 1994.
Kraus GB, Gruber RG, Knoll R., Danner U., Pharmacokinetic studies following intravenous and rectal administration of midazolam in children. Anaesthesist 38: 685–63, 1989.
Bach V., Carl P., Ravlo O., Crawford ME., Jensen AG., Mikkelsen BO., Crevoisier C., Heizmann P., Fattinger K., A randomized comparison between midazolam and thiopental for elective cesarean section: anesthesia: III. Placental transfer and elimination in neonates. Anesth Analg 68: 238–42, 1989.
Mathews HM., Carson IW, Lyons SM., Orr IA., C-upollier PS, Howard PF, Dundee JW. A Pharmacokinetic study of midazolam in paediatric patients Br j Anaesth 61: 302–7, 1988.
Jacqz — Aigrain E., Daoud P., Butrin P., Maherzi S., Beaufil F. Pharmacokinetics of midazolam during continuous infusion in critically ill neonates. Eur J Clin Pharmacol 42: 329–32, 1992.
Rey E., Delaunay L., Pons G., Murat I., Richard MO, Saint — Maurice C., Olive G. Pharmacokinetics of midazolam in children: comparative study of intranasal and intravenous administration. Eur J Clin Pharmacol 41: 355–7, 1991.
Hartwig S., Roth B., Theisohn M. Clinical experience with continuous intravenous sedation using midazolam and fentanyl in the paediatric intensive care unit. Eur J Pediatr 150: 784–8, 1991.
Tolia V., Brennan S., Aravid MK., Kauffman RE. Pharmacokinetic and pharmacodynamic study of midazolam in children during esophagogastro — duodenoscopy. J Pediatr 119:467–71, 1991.
Jacqz — Aigrain E., Wood C., Robieux I. Pharmacokinetics of midazolam in critically ill neonates. Eur J Clin Pharmacol 39: 191–2, 1990.
Jones RD., Chan K., Roulson CJ., Brown AG., Smith ID., Mya GH. Pharmacokinetics of flumazenil and midazolam. Br J Anaesth 70:286–92, 1993.
Tolia V., Brennan S., Aravind MK., Kauffman RE. Pharmacokinetic and pharmacodynamic study of midazolam in children during esophagogastroduodenoscopy. J Pediatr 119: 467–71, 1991.
Hughes J., Gill A., Mulhearn H., Powell E., Choonara I., Stedy state plasma concentration of midazolam in criticaly ill infants and children. Ann Pharmacother 30: 27–30, 1996.
Boulieu R., Lehmann B., Salord F., Fissher C., Morlet D. Pharmacokinetics of midazolam and its main metaboity 1 — hydroxymidazolam in intensive care patients. Eur J Drug Metab Pharmacokinet 23: 255–8, 1998.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nahara, M.C., McMorrow, J., Jones, P.R. et al. Pharmacokinetics of midazolam in critically ill pediatric patients. Eur. J. Drug Metab. Pharacokinet. 25, 219–221 (2000). https://doi.org/10.1007/BF03192317
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03192317